| MeSH term | MeSH ID | Detail |
|---|---|---|
| Lung Neoplasms | D008175 | 171 associated lipids |
| Body Weight | D001835 | 333 associated lipids |
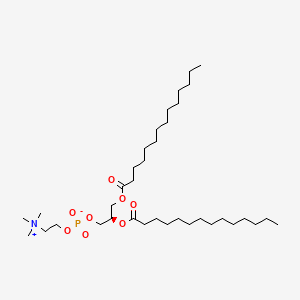
18194-24-6
18194-24-6 is a lipid of Glycerophospholipids (GP) class. 18194-24-6 is associated with abnormalities such as Cerebrovascular accident, Renal tubular disorder, Atherosclerosis, Hyperlipoproteinemia Type III and Lipid Metabolism Disorders. The involved functions are known as Process, protein folding, Catalyst, Biochemical Pathway and Fold in Medical Device Material. 18194-24-6 often locates in Tissue membrane, Membrane, periplasm, vesicle membrane and outer membrane. The associated genes with 18194-24-6 are Integral Membrane Proteins, Protein Structure, RTN4 gene, RTN4R gene and Protein, Organized by Structure. The related lipids are Micelles, dimyristoylphosphatidylglycerol, 1,2-dihexadecyl-sn-glycero-3-phosphocholine, Unilamellar Vesicles and cholesteryl oleate. The related experimental models are Mouse Model, Arthritis, Adjuvant-Induced, Disease model and Xenograft Model.
Cross Reference
Introduction
To understand associated biological information of 18194-24-6, we collected biological information of abnormalities, associated pathways, cellular/molecular locations, biological functions, related genes/proteins, lipids and common seen animal/experimental models with organized paragraphs from literatures.
What diseases are associated with 18194-24-6?
18194-24-6 is suspected in Atherosclerosis, Cardiovascular Diseases, Dehydration, Abnormal shape, Renal tubular disorder, Hyperlipoproteinemia Type III and other diseases in descending order of the highest number of associated sentences.
Related references are mostly published in these journals:
| Disease | Cross reference | Weighted score | Related literature |
|---|
Possible diseases from mapped MeSH terms on references
We collected disease MeSH terms mapped to the references associated with 18194-24-6
PubChem Associated disorders and diseases
What pathways are associated with 18194-24-6
There are no associated biomedical information in the current reference collection.
PubChem Biomolecular Interactions and Pathways
Link to PubChem Biomolecular Interactions and PathwaysWhat cellular locations are associated with 18194-24-6?
Visualization in cellular structure
Associated locations are in red color. Not associated locations are in black.
Related references are published most in these journals:
| Location | Cross reference | Weighted score | Related literatures |
|---|
What functions are associated with 18194-24-6?
Related references are published most in these journals:
| Function | Cross reference | Weighted score | Related literatures |
|---|
What lipids are associated with 18194-24-6?
Related references are published most in these journals:
| Lipid concept | Cross reference | Weighted score | Related literatures |
|---|
What genes are associated with 18194-24-6?
Related references are published most in these journals:
| Gene | Cross reference | Weighted score | Related literatures |
|---|
What common seen animal models are associated with 18194-24-6?
Mouse Model
Mouse Model are used in the study 'Association of a model class A (apolipoprotein) amphipathic alpha helical peptide with lipid: high resolution NMR studies of peptide.lipid discoidal complexes.' (Mishra VK et al., 2006).
Arthritis, Adjuvant-Induced
Arthritis, Adjuvant-Induced are used in the study 'T cell antigen receptor peptide-lipid membrane interactions using surface plasmon resonance.' (Bender V et al., 2004).
Disease model
Disease model are used in the study 'Kupffer cells do not play a role in governing the efficacy of liposomal mitoxantrone used to treat a tumor model designed to assess drug delivery to liver.' (Lim HJ et al., 2000).
Related references are published most in these journals:
| Model | Cross reference | Weighted score | Related literatures |
|---|
NCBI Entrez Crosslinks
All references with 18194-24-6
Download all related citations| Authors | Title | Published | Journal | PubMed Link |
|---|---|---|---|---|
| Wright SE and White JC | Membrane ordering effects of the anticancer agent VM-26. | 1986 | Biochim. Biophys. Acta | pmid:3024721 |
| Mims MP et al. | Motion and surface accessibility of spin-labeled lipids in a model lipoprotein containing cholesteryl oleate, dimyristoylphosphatidylcholine, and apolipoprotein E. | 1986 | Biochemistry | pmid:3026456 |
| Surewicz WK et al. | Human apolipoprotein A-I forms thermally stable complexes with anionic but not with zwitterionic phospholipids. | 1986 | J. Biol. Chem. | pmid:3097001 |
| Mantulin WW et al. | Fluorescence quenching studies of apolipoprotein A-I in solution and in lipid-protein complexes: protein dynamics. | 1986 | Biochemistry | pmid:3099838 |
| Hantz E et al. | The osmotic response of large unilamellar vesicles studied by quasielastic light scattering. | 1986 | Biochim. Biophys. Acta | pmid:3778898 |
| Dufourc EJ and Smith IC | A detailed analysis of the motions of cholesterol in biological membranes by 2H-NMR relaxation. | 1986 | Chem. Phys. Lipids | pmid:3779887 |
| Goñi FM et al. | The interaction of phosphatidylcholine bilayers with Triton X-100. | 1986 | Eur. J. Biochem. | pmid:3780729 |
| Huestis WH and Newton AC | Intermembrane protein transfer. Band 3, the erythrocyte anion transporter, transfers in native orientation from human red blood cells into the bilayer of phospholipid vesicles. | 1986 | J. Biol. Chem. | pmid:3782118 |
| Blatt E and Corin AF | The microsecond rotational motions of eosin-labelled fatty acids in multilamellar vesicles. | 1986 | Biochim. Biophys. Acta | pmid:2421777 |
| Walsh MT and Atkinson D | Reassembly of low-density lipoproteins. | 1986 | Meth. Enzymol. | pmid:3724524 |
| Cardin AD and Jackson RL | Interaction of tryptic peptides of apolipoprotein B-100 with dimyristoylphosphatidylcholine. | 1986 | Biochim. Biophys. Acta | pmid:3730406 |
| Reardon CA et al. | Expression of the human apolipoprotein E gene in cultured mammalian cells. | 1986 | J. Biol. Chem. | pmid:3733697 |
| Epand RM et al. | Structural analysis and amphiphilic properties of a chemically synthesized mitochondrial signal peptide. | 1986 | J. Biol. Chem. | pmid:3733699 |
| Walsh MT and Atkinson D | Physical properties of apoprotein B in mixed micelles with sodium deoxycholate and in a vesicle with dimyristoyl phosphatidylcholine. | 1986 | J. Lipid Res. | pmid:3734628 |
| Nakagaki M et al. | [Action of phospholipase D on the L-alpha-dimyristoyl phosphatidylcholine/cholesterol liposome]. | 1986 | Yakugaku Zasshi | pmid:3712235 |
| Jacobs RE and White SH | Mixtures of a series of homologous hydrophobic peptides with lipid bilayers: a simple model system for examining the protein-lipid interface. | 1986 | Biochemistry | pmid:3718968 |
| Hermetter A and Lakowicz JR | The aggregation state of mellitin in lipid bilayers. An energy transfer study. | 1986 | J. Biol. Chem. | pmid:3722154 |
| Mims MP et al. | Microemulsions of cholesteryl oleate and dimyristoylphosphatidylcholine: a model for cholesteryl ester rich very low density lipoproteins. | 1986 | Biochemistry | pmid:3456798 |
| Griffin EA et al. | Anthracycline binding to synthetic and natural membranes. A study using resonance energy transfer. | 1986 | Biochemistry | pmid:3467794 |
| Caughey B et al. | The role of molecular conformation in ion capture by carboxylic ionophores: a circular dichroism study of narasin A in single-phase solvents and liposomes. | 1986 | Biochim. Biophys. Acta | pmid:3002459 |