| MeSH term | MeSH ID | Detail |
|---|---|---|
| Blastomycosis | D001759 | 5 associated lipids |
| Lymphoma, Primary Effusion | D054685 | 2 associated lipids |
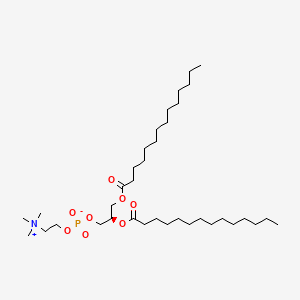
18194-24-6
18194-24-6 is a lipid of Glycerophospholipids (GP) class. 18194-24-6 is associated with abnormalities such as Cerebrovascular accident, Renal tubular disorder, Atherosclerosis, Hyperlipoproteinemia Type III and Lipid Metabolism Disorders. The involved functions are known as Process, protein folding, Catalyst, Biochemical Pathway and Fold in Medical Device Material. 18194-24-6 often locates in Tissue membrane, Membrane, periplasm, vesicle membrane and outer membrane. The associated genes with 18194-24-6 are Integral Membrane Proteins, Protein Structure, RTN4 gene, RTN4R gene and Protein, Organized by Structure. The related lipids are Micelles, dimyristoylphosphatidylglycerol, 1,2-dihexadecyl-sn-glycero-3-phosphocholine, Unilamellar Vesicles and cholesteryl oleate. The related experimental models are Mouse Model, Arthritis, Adjuvant-Induced, Disease model and Xenograft Model.
Cross Reference
Introduction
To understand associated biological information of 18194-24-6, we collected biological information of abnormalities, associated pathways, cellular/molecular locations, biological functions, related genes/proteins, lipids and common seen animal/experimental models with organized paragraphs from literatures.
What diseases are associated with 18194-24-6?
18194-24-6 is suspected in Atherosclerosis, Cardiovascular Diseases, Dehydration, Abnormal shape, Renal tubular disorder, Hyperlipoproteinemia Type III and other diseases in descending order of the highest number of associated sentences.
Related references are mostly published in these journals:
| Disease | Cross reference | Weighted score | Related literature |
|---|
Possible diseases from mapped MeSH terms on references
We collected disease MeSH terms mapped to the references associated with 18194-24-6
PubChem Associated disorders and diseases
What pathways are associated with 18194-24-6
There are no associated biomedical information in the current reference collection.
PubChem Biomolecular Interactions and Pathways
Link to PubChem Biomolecular Interactions and PathwaysWhat cellular locations are associated with 18194-24-6?
Visualization in cellular structure
Associated locations are in red color. Not associated locations are in black.
Related references are published most in these journals:
| Location | Cross reference | Weighted score | Related literatures |
|---|
What functions are associated with 18194-24-6?
Related references are published most in these journals:
| Function | Cross reference | Weighted score | Related literatures |
|---|
What lipids are associated with 18194-24-6?
Related references are published most in these journals:
| Lipid concept | Cross reference | Weighted score | Related literatures |
|---|
What genes are associated with 18194-24-6?
Related references are published most in these journals:
| Gene | Cross reference | Weighted score | Related literatures |
|---|
What common seen animal models are associated with 18194-24-6?
Mouse Model
Mouse Model are used in the study 'Association of a model class A (apolipoprotein) amphipathic alpha helical peptide with lipid: high resolution NMR studies of peptide.lipid discoidal complexes.' (Mishra VK et al., 2006).
Arthritis, Adjuvant-Induced
Arthritis, Adjuvant-Induced are used in the study 'T cell antigen receptor peptide-lipid membrane interactions using surface plasmon resonance.' (Bender V et al., 2004).
Disease model
Disease model are used in the study 'Kupffer cells do not play a role in governing the efficacy of liposomal mitoxantrone used to treat a tumor model designed to assess drug delivery to liver.' (Lim HJ et al., 2000).
Related references are published most in these journals:
| Model | Cross reference | Weighted score | Related literatures |
|---|
NCBI Entrez Crosslinks
All references with 18194-24-6
Download all related citations| Authors | Title | Published | Journal | PubMed Link |
|---|---|---|---|---|
| Bian J et al. | Solid-phase extraction approach for phospholipids profiling by titania-coated silica microspheres prior to reversed-phase liquid chromatography-evaporative light scattering detection and tandem mass spectrometry analysis. | 2014 | Talanta | pmid:24725887 |
| OlguÃn Y et al. | The effects of lipids and surfactants on TLR5-proteoliposome functionality for flagellin detection using surface plasmon resonance biosensing. | 2014 | Talanta | pmid:24881544 |
| Baday S et al. | Mechanism of NH4(+) Recruitment and NH3 Transport in Rh Proteins. | 2015 | Structure | pmid:26190573 |
| Chen W et al. | The Unusual Transmembrane Partition of the Hexameric Channel of the Hepatitis C Virus. | 2018 | Structure | pmid:29551287 |
| Murphy RE et al. | Solution Structure and Membrane Interaction of the Cytoplasmic Tail of HIV-1 gp41 Protein. | 2017 | Structure | pmid:29056482 |
| Zachariae U et al. | β-Barrel mobility underlies closure of the voltage-dependent anion channel. | 2012 | Structure | pmid:22841291 |
| Holyoake J and Sansom MS | Conformational change in an MFS protein: MD simulations of LacY. | 2007 | Structure | pmid:17637346 |
| Byrne C et al. | ERα17p, a peptide reproducing the hinge region of the estrogen receptor α associates to biological membranes: A biophysical approach. | 2012 | Steroids | pmid:22426414 |
| Li GC et al. | Solid-state NMR analysis of steroidal conformation of 17α- and 17β-estradiol in the absence and presence of lipid environment. | 2012 | Steroids | pmid:22155023 |
| Conaughty JM et al. | Antifungal penetration into normal rabbit nucleus pulposus. | 2004 | Spine | pmid:15247589 |