| MeSH term | MeSH ID | Detail |
|---|---|---|
| Porcine Reproductive and Respiratory Syndrome | D019318 | 4 associated lipids |
| Cicatrix, Hypertrophic | D017439 | 4 associated lipids |
| Spinocerebellar Ataxias | D020754 | 4 associated lipids |
| Nasopharyngeal Neoplasms | D009303 | 4 associated lipids |
| Neuroendocrine Tumors | D018358 | 4 associated lipids |
| Myeloproliferative Disorders | D009196 | 5 associated lipids |
| Opioid-Related Disorders | D009293 | 5 associated lipids |
| Osteomalacia | D010018 | 5 associated lipids |
| Fragile X Syndrome | D005600 | 5 associated lipids |
| Carcinoma, Pancreatic Ductal | D021441 | 6 associated lipids |
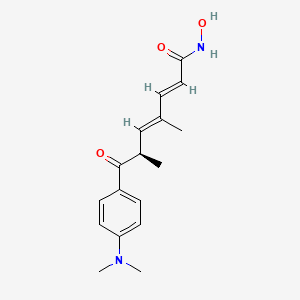
trichostatin A
Trichostatin is a lipid of Polyketides (PK) class. Trichostatin is associated with abnormalities such as Dentatorubral-Pallidoluysian Atrophy, PARAGANGLIOMAS 3, abnormal fragmented structure, Disintegration (morphologic abnormality) and Hyperostosis, Diffuse Idiopathic Skeletal. The involved functions are known as Acetylation, Cell Differentiation process, histone modification, Gene Silencing and Transcriptional Activation. Trichostatin often locates in CD41a, Hematopoietic System, Chromatin Structure, Blood and Endothelium. The associated genes with Trichostatin are SPI1 gene, CELL Gene, Chromatin, CXCR4 gene and DNMT1 gene. The related lipids are Butyrates, Promega, butyrate, Lipopolysaccharides and Steroids. The related experimental models are Knock-out, Mouse Model, Xenograft Model and Cancer Model.
Cross Reference
Introduction
To understand associated biological information of trichostatin A, we collected biological information of abnormalities, associated pathways, cellular/molecular locations, biological functions, related genes/proteins, lipids and common seen animal/experimental models with organized paragraphs from literatures.
What diseases are associated with trichostatin A?
trichostatin A is suspected in Infection, Morphologically altered structure, Ureteral obstruction, Photosensitization, Atherosclerosis, Hypertrophic Cardiomyopathy and other diseases in descending order of the highest number of associated sentences.
Related references are mostly published in these journals:
| Disease | Cross reference | Weighted score | Related literature |
|---|
Possible diseases from mapped MeSH terms on references
We collected disease MeSH terms mapped to the references associated with trichostatin A
PubChem Associated disorders and diseases
What pathways are associated with trichostatin A
Lipid pathways are not clear in current pathway databases. We organized associated pathways with trichostatin A through full-text articles, including metabolic pathways or pathways of biological mechanisms.
Related references are published most in these journals:
| Pathway name | Related literatures |
|---|
PubChem Biomolecular Interactions and Pathways
Link to PubChem Biomolecular Interactions and PathwaysWhat cellular locations are associated with trichostatin A?
Visualization in cellular structure
Associated locations are in red color. Not associated locations are in black.
Related references are published most in these journals:
| Location | Cross reference | Weighted score | Related literatures |
|---|
What functions are associated with trichostatin A?
Related references are published most in these journals:
| Function | Cross reference | Weighted score | Related literatures |
|---|
What lipids are associated with trichostatin A?
Related references are published most in these journals:
| Lipid concept | Cross reference | Weighted score | Related literatures |
|---|
What genes are associated with trichostatin A?
Related references are published most in these journals:
| Gene | Cross reference | Weighted score | Related literatures |
|---|
What common seen animal models are associated with trichostatin A?
Mouse Model
Mouse Model are used in the study 'Regulation of minichromosome maintenance gene family by microRNA-1296 and genistein in prostate cancer.' (Majid S et al., 2010), Mouse Model are used in the study 'Reversal of hypermethylation and reactivation of p16INK4a, RARbeta, and MGMT genes by genistein and other isoflavones from soy.' (Fang MZ et al., 2005) and Mouse Model are used in the study 'Histone deacetylase 3 mediates allergic skin inflammation by regulating expression of MCP1 protein.' (Kim Y et al., 2012).
Xenograft Model
Xenograft Model are used in the study 'Histone deacetylase inhibitors induce growth arrest and differentiation in uveal melanoma.' (Landreville S et al., 2012), Xenograft Model are used in the study 'Extended treatment with physiologic concentrations of dietary phytochemicals results in altered gene expression, reduced growth, and apoptosis of cancer cells.' (Moiseeva EP et al., 2007) and Xenograft Model are used in the study 'Retinoic acid and the histone deacetylase inhibitor trichostatin a inhibit the proliferation of human renal cell carcinoma in a xenograft tumor model.' (Touma SE et al., 2005).
Cancer Model
Cancer Model are used in the study 'Plasma pharmacokinetics and metabolism of the histone deacetylase inhibitor trichostatin a after intraperitoneal administration to mice.' (Sanderson L et al., 2004).
Related references are published most in these journals:
| Model | Cross reference | Weighted score | Related literatures |
|---|
NCBI Entrez Crosslinks
All references with trichostatin A
Download all related citations| Authors | Title | Published | Journal | PubMed Link |
|---|---|---|---|---|
| Lavoie R et al. | Design and synthesis of a novel class of histone deacetylase inhibitors. | 2001 | Bioorg. Med. Chem. Lett. | pmid:11597413 |
| Uesato S et al. | Novel histone deacetylase inhibitors: N-hydroxycarboxamides possessing a terminal bicyclic aryl group. | 2002 | Bioorg. Med. Chem. Lett. | pmid:11992774 |
| Van Ommeslaeghe K et al. | Amide analogues of TSA: synthesis, binding mode analysis and HDAC inhibition. | 2003 | Bioorg. Med. Chem. Lett. | pmid:12749885 |
| Vasudevan A et al. | Heterocyclic ketones as inhibitors of histone deacetylase. | 2003 | Bioorg. Med. Chem. Lett. | pmid:14592473 |
| Vaisburg A et al. | (2-amino-phenyl)-amides of omega-substituted alkanoic acids as new histone deacetylase inhibitors. | 2004 | Bioorg. Med. Chem. Lett. | pmid:14684344 |
| Wang DF et al. | QSAR studies of PC-3 cell line inhibition activity of TSA and SAHA-like hydroxamic acids. | 2004 | Bioorg. Med. Chem. Lett. | pmid:14741273 |
| Pabba C et al. | Design and synthesis of aryl ether and sulfone hydroxamic acids as potent histone deacetylase (HDAC) inhibitors. | 2011 | Bioorg. Med. Chem. Lett. | pmid:21109435 |
| Giannini G et al. | N-Hydroxy-(4-oxime)-cinnamide: a versatile scaffold for the synthesis of novel histone deacetylase [correction of deacetilase] (HDAC) inhibitors. | 2009 | Bioorg. Med. Chem. Lett. | pmid:19285395 |
| Riester D et al. | Non-isotopic dual parameter competition assay suitable for high-throughput screening of histone deacetylases. | 2009 | Bioorg. Med. Chem. Lett. | pmid:19457659 |
| Desai D et al. | SelSA, selenium analogs of SAHA as potent histone deacetylase inhibitors. | 2010 | Bioorg. Med. Chem. Lett. | pmid:20167479 |
| Gupta PK et al. | Inhibitors selective for HDAC6 in enzymes and cells. | 2010 | Bioorg. Med. Chem. Lett. | pmid:20947351 |
| Yu D et al. | Clonal Rett Syndrome cell lines to test compounds for activation of wild-type MeCP2 expression. | 2011 | Bioorg. Med. Chem. Lett. | pmid:21840716 |
| Charrier C et al. | Antiproliferative activities of a library of hybrids between indanones and HDAC inhibitor SAHA and MS-275 analogues. | 2007 | Bioorg. Med. Chem. Lett. | pmid:17897824 |
| Suzuki T et al. | Identification of a potent and stable antiproliferative agent by the prodrug formation of a thiolate histone deacetylase inhibitor. | 2007 | Bioorg. Med. Chem. Lett. | pmid:17257837 |
| Lee S et al. | Design, synthesis, and evaluation of isoindolinone-hydroxamic acid derivatives as histone deacetylase (HDAC) inhibitors. | 2007 | Bioorg. Med. Chem. Lett. | pmid:17588744 |
| Loudni L et al. | Design, synthesis and biological evaluation of 1,4-benzodiazepine-2,5-dione-based HDAC inhibitors. | 2007 | Bioorg. Med. Chem. Lett. | pmid:17624773 |
| Charrier C et al. | Synthesis of rigid trichostatin A analogs as HDAC inhibitors. | 2006 | Bioorg. Med. Chem. Lett. | pmid:16904890 |
| Shinji C et al. | Design and synthesis of phthalimide-type histone deacetylase inhibitors. | 2005 | Bioorg. Med. Chem. Lett. | pmid:16137884 |
| Hirata Y et al. | Anti-tumor activity of new orally bioavailable 2-amino-5-(thiophen-2-yl)benzamide-series histone deacetylase inhibitors, possessing an aqueous soluble functional group as a surface recognition domain. | 2012 | Bioorg. Med. Chem. Lett. | pmid:22321215 |
| Liao V et al. | Amide-based derivatives of β-alanine hydroxamic acid as histone deacetylase inhibitors: attenuation of potency through resonance effects. | 2012 | Bioorg. Med. Chem. Lett. | pmid:22932316 |