| MeSH term | MeSH ID | Detail |
|---|---|---|
| Epididymitis | D004823 | 1 associated lipids |
| Pruritus Vulvae | D011539 | 1 associated lipids |
| Central Serous Chorioretinopathy | D056833 | 1 associated lipids |
| Feline Acquired Immunodeficiency Syndrome | D016181 | 1 associated lipids |
| Budd-Chiari Syndrome | D006502 | 1 associated lipids |
| AIDS-Associated Nephropathy | D016263 | 1 associated lipids |
| Lentigo | D007911 | 1 associated lipids |
| Mutism | D009155 | 1 associated lipids |
| Amyloid Neuropathies | D017772 | 1 associated lipids |
| Cervical Intraepithelial Neoplasia | D018290 | 1 associated lipids |
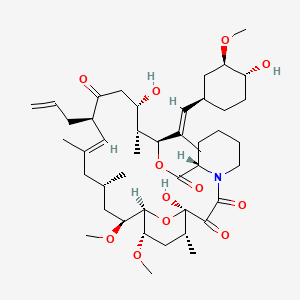
tacrolimus
Tacrolimus is a lipid of Polyketides (PK) class. Tacrolimus is associated with abnormalities such as Renal glomerular disease. The involved functions are known as inhibitors, Fungicidal activity, Metabolic Inhibition, Excretory function and Dephosphorylation. Tacrolimus often locates in Hepatic, Mitochondrial matrix and Inner mitochondrial membrane. The associated genes with Tacrolimus are RHOA gene and BGN gene.
Cross Reference
Introduction
To understand associated biological information of tacrolimus, we collected biological information of abnormalities, associated pathways, cellular/molecular locations, biological functions, related genes/proteins, lipids and common seen animal/experimental models with organized paragraphs from literatures.
What diseases are associated with tacrolimus?
tacrolimus is suspected in Renal glomerular disease, Candidiasis, Mycoses, PARKINSON DISEASE, LATE-ONSET, Morphologically altered structure, Skin Diseases, Infectious and other diseases in descending order of the highest number of associated sentences.
Related references are mostly published in these journals:
- Antimicrob. Agents Chemother. (2)
- Am. J. Physiol. Renal Physiol. (1)
- Drug Metab. Dispos. (1)
- Others (1)
| Disease | Cross reference | Weighted score | Related literature |
|---|
Possible diseases from mapped MeSH terms on references
We collected disease MeSH terms mapped to the references associated with tacrolimus
PubChem Associated disorders and diseases
What pathways are associated with tacrolimus
There are no associated biomedical information in the current reference collection.
PubChem Biomolecular Interactions and Pathways
Link to PubChem Biomolecular Interactions and PathwaysWhat cellular locations are associated with tacrolimus?
Visualization in cellular structure
Associated locations are in red color. Not associated locations are in black.
Related references are published most in these journals:
| Location | Cross reference | Weighted score | Related literatures |
|---|
What functions are associated with tacrolimus?
Related references are published most in these journals:
| Function | Cross reference | Weighted score | Related literatures |
|---|
What lipids are associated with tacrolimus?
Related references are published most in these journals:
| Lipid concept | Cross reference | Weighted score | Related literatures |
|---|
What genes are associated with tacrolimus?
Related references are published most in these journals:
| Gene | Cross reference | Weighted score | Related literatures |
|---|
What common seen animal models are associated with tacrolimus?
There are no associated biomedical information in the current reference collection.
NCBI Entrez Crosslinks
All references with tacrolimus
Download all related citations| Authors | Title | Published | Journal | PubMed Link |
|---|---|---|---|---|
| Wiebe C et al. | Class II Eplet Mismatch Modulates Tacrolimus Trough Levels Required to Prevent Donor-Specific Antibody Development. | 2017 | J. Am. Soc. Nephrol. | pmid:28729289 |
| Tolkachjov SN et al. | Frontal fibrosing alopecia among men: AÂ clinicopathologic study of 7 cases. | 2017 | J. Am. Acad. Dermatol. | pmid:28716436 |
| Montenovo MI et al. | Superior Patient and Graft Survival in Adult Liver Transplant with Rabbit Antithymocyte Globulin Induction: Experience with 595 Patients. | 2017 | Exp Clin Transplant | pmid:27309029 |
| Das B et al. | Alemtuzumab (Campath-1H) therapy for refractory rejections in pediatric heart transplant recipients. | 2017 | Pediatr Transplant | pmid:27862703 |
| Wu Z et al. | FOXP3 rs3761548 polymorphism is associated with tacrolimus-induced acute nephrotoxicity in renal transplant patients. | 2017 | Eur. J. Clin. Pharmacol. | pmid:27747372 |
| Tamashiro EY et al. | Influence of CYP3A4 and CYP3A5 polymorphisms on tacrolimus and sirolimus exposure in stable kidney transplant recipients. | 2017 | Drug Metab Pers Ther | pmid:28593920 |
| Fai D et al. | Duration of response in vitiligo lesions after narrowband UVB phototherapy combined with tacrolimus ointment. | 2017 | G Ital Dermatol Venereol | pmid:28621124 |
| Nakagawa K et al. | Immunosuppressive treatment using tacrolimus promotes pregnancy outcome in infertile women with repeated implantation failures. | 2017 | Am. J. Reprod. Immunol. | pmid:28466977 |
| Ramirez R et al. | Posterior Reversible Encephalopathy Syndrome After Orthotopic Heart Transplantation: A Case Report. | 2017 | Am J Case Rep | pmid:28465499 |
| Harada S et al. | Complete remission of angiolymphoid hyperplasia with eosinophilia using topical tacrolimus. | 2017 | Eur J Dermatol | pmid:28007668 |
| Wan T et al. | Effects of nanoparticles with hydrotropic nicotinamide on tacrolimus: permeability through psoriatic skin and antipsoriatic and antiproliferative activities. | 2017 | Int J Nanomedicine | pmid:28260894 |
| Cui W et al. | Therapy of tacrolimus combined with corticosteroids in idiopathic membranous nephropathy. | 2017 | Braz. J. Med. Biol. Res. | pmid:28355356 |
| Tremblay S et al. | A Steady-State Head-to-Head Pharmacokinetic Comparison of All FK-506 (Tacrolimus) Formulations (ASTCOFF): An Open-Label, Prospective, Randomized, Two-Arm, Three-Period Crossover Study. | 2017 | Am. J. Transplant. | pmid:27340950 |
| Vanhove T et al. | Response to: 'Bodyweight-adjustments introduce significant correlations between CYP3A metrics and tacrolimus clearance'. | 2017 | Br J Clin Pharmacol | pmid:28168728 |
| Qin HZ et al. | Evaluating tacrolimus treatment in idiopathic membranous nephropathy in a cohort of 408 patients. | 2017 | BMC Nephrol | pmid:28056860 |
| Kim YK et al. | New tablet formulation of tacrolimus with smaller interindividual variability may become a better treatment option than the conventional capsule formulation in organ transplant patients. | 2017 | Drug Des Devel Ther | pmid:29033547 |
| Dutta D et al. | Recruitment of calcineurin to the TCR positively regulates T cell activation. | 2017 | Nat. Immunol. | pmid:27941787 |
| Terakura S et al. | Exploratory research for optimal GvHD prophylaxis after single unit CBT in adults: short-term methotrexate reduced the incidence of severe GvHD more than mycophenolate mofetil. | 2017 | Bone Marrow Transplant. | pmid:27941766 |
| Naicker D et al. | Nationwide conversion to generic tacrolimus in pediatric kidney transplant recipients. | 2017 | Pediatr. Nephrol. | pmid:28660366 |
| Kannegieter NM et al. | Pharmacodynamic Monitoring of Tacrolimus-Based Immunosuppression in CD14+ Monocytes After Kidney Transplantation. | 2017 | Ther Drug Monit | pmid:28640063 |