| MeSH term | MeSH ID | Detail |
|---|---|---|
| Diabetes Mellitus, Type 1 | D003922 | 56 associated lipids |
| Alzheimer Disease | D000544 | 76 associated lipids |
| Colonic Neoplasms | D003110 | 161 associated lipids |
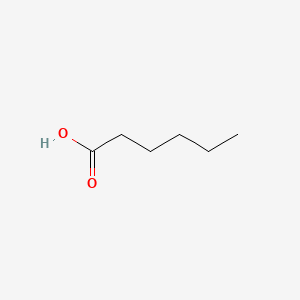
HEXANOIC ACID
HEXANOIC ACID is a lipid of Fatty Acyls (FA) class. Hexanoic acid is associated with abnormalities such as Obesity, Ileoanal Pouches, Ulcerative Colitis, Ulcerative colitis, quiescent and Diabetes Mellitus, Non-Insulin-Dependent. The involved functions are known as Binding (Molecular Function), Anabolism, Adjudication, enzyme activity and Process. Hexanoic acid often locates in Membrane, Tissue membrane, Microsomes, Liver, Microsomes and Mitochondria. The associated genes with HEXANOIC ACID are SH2D1A gene, Fusion Protein and MAPK3 gene. The related lipids are hexanoic acid, Fatty Acids, Butyric Acid, Propionate and Palmitates.
Cross Reference
Introduction
To understand associated biological information of HEXANOIC ACID, we collected biological information of abnormalities, associated pathways, cellular/molecular locations, biological functions, related genes/proteins, lipids and common seen animal/experimental models with organized paragraphs from literatures.
What diseases are associated with HEXANOIC ACID?
HEXANOIC ACID is suspected in Obesity, Ileoanal Pouches, Ulcerative Colitis, Ulcerative colitis, quiescent, Diabetes Mellitus, Non-Insulin-Dependent, Gastroesophageal reflux disease and other diseases in descending order of the highest number of associated sentences.
Related references are mostly published in these journals:
| Disease | Cross reference | Weighted score | Related literature |
|---|
Possible diseases from mapped MeSH terms on references
We collected disease MeSH terms mapped to the references associated with HEXANOIC ACID
PubChem Associated disorders and diseases
What pathways are associated with HEXANOIC ACID
There are no associated biomedical information in the current reference collection.
PubChem Biomolecular Interactions and Pathways
Link to PubChem Biomolecular Interactions and PathwaysWhat cellular locations are associated with HEXANOIC ACID?
Visualization in cellular structure
Associated locations are in red color. Not associated locations are in black.
Related references are published most in these journals:
| Location | Cross reference | Weighted score | Related literatures |
|---|
What functions are associated with HEXANOIC ACID?
Related references are published most in these journals:
| Function | Cross reference | Weighted score | Related literatures |
|---|
What lipids are associated with HEXANOIC ACID?
Related references are published most in these journals:
| Lipid concept | Cross reference | Weighted score | Related literatures |
|---|
What genes are associated with HEXANOIC ACID?
Related references are published most in these journals:
| Gene | Cross reference | Weighted score | Related literatures |
|---|
What common seen animal models are associated with HEXANOIC ACID?
There are no associated biomedical information in the current reference collection.
NCBI Entrez Crosslinks
All references with HEXANOIC ACID
Download all related citations| Authors | Title | Published | Journal | PubMed Link |
|---|---|---|---|---|
| Zhao G et al. | Rapid determination of short-chain fatty acids in colonic contents and faeces of humans and rats by acidified water-extraction and direct-injection gas chromatography. | 2006 | Biomed. Chromatogr. | pmid:16206138 |
| Chalmers RA and Lawson AM | Identification of 5-hydroxyhexanoic acid in the urine of twin siblings with a Reye's-like syndrome associated with dicarboxylic aciduria and hypoglycaemia and with similarities to Jamaican vomiting sickness. | 1979 | Biomed. Mass Spectrom. | pmid:575058 |
| Dokolina EV et al. | [Semisynthetic cerebrosides]. | 1983 | Bioorg. Khim. | pmid:6680305 |
| Greiner C et al. | Identification of 2-mercaptohexanoic acids as dual inhibitors of 5-lipoxygenase and microsomal prostaglandin Eâ‚‚ synthase-1. | 2011 | Bioorg. Med. Chem. | pmid:21570310 |
| Ferrari AM et al. | Validation of an automated procedure for the prediction of relative free energies of binding on a set of aldose reductase inhibitors. | 2007 | Bioorg. Med. Chem. | pmid:17870536 |
| Wang Y et al. | Synthesis and antitumor activity evaluations of albumin-binding prodrugs of CC-1065 analog. | 2008 | Bioorg. Med. Chem. | pmid:18508273 |
| Spickenreither M et al. | Novel 6-O-acylated vitamin C derivatives as hyaluronidase inhibitors with selectivity for bacterial lyases. | 2006 | Bioorg. Med. Chem. Lett. | pmid:16908142 |
| Flesch D et al. | SAR-studies of γ-secretase modulators with PPARγ-agonistic and 5-lipoxygenase-inhibitory activity for Alzheimer's disease. | 2015 | Bioorg. Med. Chem. Lett. | pmid:25575659 |
| Li S et al. | Imaging molecular transport across lipid bilayers. | 2011 | Biophys. J. | pmid:21806938 |
| Liu Y et al. | Alcohol-to-acid ratio and substrate concentration affect product structure in chain elongation reactions initiated by unacclimatized inoculum. | 2016 | Bioresour. Technol. | pmid:27469095 |