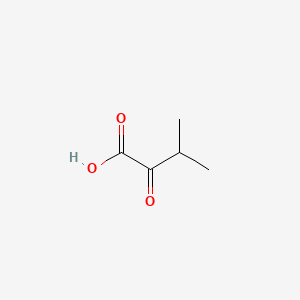
3-Methyl-2-oxobutanoic acid
3-Methyl-2-oxobutanoic acid is a lipid of Fatty Acyls (FA) class. 3-methyl-2-oxobutanoic acid is associated with abnormalities such as Maple Syrup Urine Disease and Kidney Failure, Chronic. The involved functions are known as Phosphorylation, Citric Acid Cycle, inhibitors, Process and Metabolic Control. 3-methyl-2-oxobutanoic acid often locates in Mitochondria, BL21, Cytoplasm, Ribosomes and Head. The associated genes with 3-Methyl-2-oxobutanoic acid are Genome, Homologous Gene, Operon, Alleles and Oxidoreductase Gene. The related lipids are dimyristoylphosphatidylglycerol, 9-oxononanoic acid, Valerates and alpha-ketocaproic acid.
Cross Reference
Introduction
To understand associated biological information of 3-Methyl-2-oxobutanoic acid, we collected biological information of abnormalities, associated pathways, cellular/molecular locations, biological functions, related genes/proteins, lipids and common seen animal/experimental models with organized paragraphs from literatures.
What diseases are associated with 3-Methyl-2-oxobutanoic acid?
3-Methyl-2-oxobutanoic acid is suspected in Maple Syrup Urine Disease, Kidney Failure, Chronic and other diseases in descending order of the highest number of associated sentences.
Related references are mostly published in these journals:
| Disease | Cross reference | Weighted score | Related literature |
|---|
Possible diseases from mapped MeSH terms on references
We collected disease MeSH terms mapped to the references associated with 3-Methyl-2-oxobutanoic acid
PubChem Associated disorders and diseases
What pathways are associated with 3-Methyl-2-oxobutanoic acid
There are no associated biomedical information in the current reference collection.
PubChem Biomolecular Interactions and Pathways
Link to PubChem Biomolecular Interactions and PathwaysWhat cellular locations are associated with 3-Methyl-2-oxobutanoic acid?
Visualization in cellular structure
Associated locations are in red color. Not associated locations are in black.
Related references are published most in these journals:
| Location | Cross reference | Weighted score | Related literatures |
|---|
What functions are associated with 3-Methyl-2-oxobutanoic acid?
Related references are published most in these journals:
| Function | Cross reference | Weighted score | Related literatures |
|---|
What lipids are associated with 3-Methyl-2-oxobutanoic acid?
Related references are published most in these journals:
| Lipid concept | Cross reference | Weighted score | Related literatures |
|---|
What genes are associated with 3-Methyl-2-oxobutanoic acid?
Related references are published most in these journals:
| Gene | Cross reference | Weighted score | Related literatures |
|---|
What common seen animal models are associated with 3-Methyl-2-oxobutanoic acid?
There are no associated biomedical information in the current reference collection.
NCBI Entrez Crosslinks
All references with 3-Methyl-2-oxobutanoic acid
Download all related citations| Authors | Title | Published | Journal | PubMed Link |
|---|---|---|---|---|
| pmid: | ||||
| Puukka M et al. | Formation of 2-oxoisovalerate dehydrogenase in Pseudomonas fluorescens. | 1972 | Acta Chem Scand | pmid:4628011 |
| Brodelius P | High-performance liquid chromatographic analysis of analogous amino and oxo acids for the determination of amino acid oxidase and transaminase activities. | 1984 | Acta Chem. Scand., B, Org. Chem. Biochem. | pmid:6147057 |
| Koon N et al. | Crystallization and preliminary X-ray analysis of alpha-isopropylmalate synthase from Mycobacterium tuberculosis. | 2004 | Acta Crystallogr. D Biol. Crystallogr. | pmid:15159590 |
| Berthold CL et al. | Structure of the branched-chain keto acid decarboxylase (KdcA) from Lactococcus lactis provides insights into the structural basis for the chemoselective and enantioselective carboligation reaction. | 2007 | Acta Crystallogr. D Biol. Crystallogr. | pmid:18084069 |
| Petit-Haertlein I et al. | Incorporation of methyl-protonated valine and leucine residues into deuterated ocean pout type III antifreeze protein: expression, crystallization and preliminary neutron diffraction studies. | 2010 | Acta Crystallogr. Sect. F Struct. Biol. Cryst. Commun. | pmid:20516595 |
| Takei N | Branched chain amino acid transaminase and branched chain alpha-ketoacid dehydrogenase activity in the brain, liver and skeletal muscle of acute hepatic failure rats. | 1985 | Acta Med. Okayama | pmid:3984777 |
| Gerez de Burgos NM et al. | [Alpha-hydroxy-acid dehydrogenase activity in Trypanosoma cruzi]. | 1976 | Acta Physiol Lat Am | pmid:801103 |
| WRETLIND KA | Replacement of valine in the diet by alpha-ketoisovaleric acid. | 1952 | Acta Physiol. Scand. | pmid:13050479 |
| Barve A et al. | A kinetic platform for in silico modeling of the metabolic dynamics in Escherichia coli. | 2010 | Adv Appl Bioinform Chem | pmid:21918631 |