| MeSH term | MeSH ID | Detail |
|---|---|---|
| Leukemia, Hairy Cell | D007943 | 5 associated lipids |
| Abetalipoproteinemia | D000012 | 7 associated lipids |
| Xanthomatosis | D014973 | 17 associated lipids |
| Carcinoma, Hepatocellular | D006528 | 140 associated lipids |
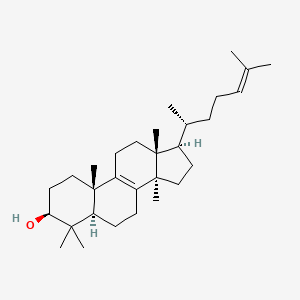
lanosterol
lanosterol is a lipid of Sterol Lipids (ST) class. Lanosterol is associated with abnormalities such as Infection, Atherosclerosis, Myocardial Infarction, Chagas Disease and Fatty Liver. The involved functions are known as Signal, Cytokinesis, physiological aspects, Stereochemistry and ergosterol biosynthetic process. Lanosterol often locates in Body tissue, Membrane, Plasma membrane, Tissue membrane and Cytoskeletal Filaments. The associated genes with lanosterol are Retinoic Acid Response Element, P4HTM gene, CYP51A1 gene, HM13 gene and SC4MOL gene. The related lipids are pneumocysterol, Sterols, lanosteryl acetate, ebericol and cycloartenol. The related experimental models are Knock-out.
Cross Reference
Introduction
To understand associated biological information of lanosterol, we collected biological information of abnormalities, associated pathways, cellular/molecular locations, biological functions, related genes/proteins, lipids and common seen animal/experimental models with organized paragraphs from literatures.
What diseases are associated with lanosterol?
lanosterol is suspected in Atherosclerosis, Chagas Disease, vaginalis, hypercholesterolemia, Obesity, Infection and other diseases in descending order of the highest number of associated sentences.
Related references are mostly published in these journals:
| Disease | Cross reference | Weighted score | Related literature |
|---|
Possible diseases from mapped MeSH terms on references
We collected disease MeSH terms mapped to the references associated with lanosterol
PubChem Associated disorders and diseases
What pathways are associated with lanosterol
Lipid pathways are not clear in current pathway databases. We organized associated pathways with lanosterol through full-text articles, including metabolic pathways or pathways of biological mechanisms.
Related references are published most in these journals:
| Pathway name | Related literatures |
|---|
PubChem Biomolecular Interactions and Pathways
Link to PubChem Biomolecular Interactions and PathwaysWhat cellular locations are associated with lanosterol?
Visualization in cellular structure
Associated locations are in red color. Not associated locations are in black.
Related references are published most in these journals:
| Location | Cross reference | Weighted score | Related literatures |
|---|
What functions are associated with lanosterol?
Related references are published most in these journals:
| Function | Cross reference | Weighted score | Related literatures |
|---|
What lipids are associated with lanosterol?
Related references are published most in these journals:
| Lipid concept | Cross reference | Weighted score | Related literatures |
|---|
What genes are associated with lanosterol?
Related references are published most in these journals:
| Gene | Cross reference | Weighted score | Related literatures |
|---|
What common seen animal models are associated with lanosterol?
Knock-out
Knock-out are used in the study 'Dual biosynthetic pathways to phytosterol via cycloartenol and lanosterol in Arabidopsis.' (Ohyama K et al., 2009), Knock-out are used in the study 'Expression, purification, and characterization of Aspergillus fumigatus sterol 14-alpha demethylase (CYP51) isoenzymes A and B.' (Warrilow AG et al., 2010) and Knock-out are used in the study 'Potential biological functions of cytochrome P450 reductase-dependent enzymes in small intestine: novel link to expression of major histocompatibility complex class II genes.' (D'Agostino J et al., 2012).
Related references are published most in these journals:
| Model | Cross reference | Weighted score | Related literatures |
|---|
NCBI Entrez Crosslinks
All references with lanosterol
Download all related citations| Authors | Title | Published | Journal | PubMed Link |
|---|---|---|---|---|
| Visbal G et al. | Synthesis, in vitro antifungal activity and mechanism of action of four sterol hydrazone analogues against the dimorphic fungus Paracoccidioides brasiliensis. | Steroids | pmid:21605581 | |
| Skinner C and Miraldi Utz V | Pharmacological approaches to restoring lens transparency: Real world applications. | Ophthalmic Genet. | pmid:27648776 | |
| pmid: | ||||
| DOREE C et al. | Lanosterol; hydrocarbons formed by the action of dehydrating agents. | 1947 | J Chem Soc | pmid:18920806 |
| DOREE C et al. | Position of the hydroxyl group in lanosterol. | 1949 | Nature | pmid:18107580 |
| MARCUS S | Antibacterial activity of the triterpenoid acid (polyporenic acid C) and of ungulinic acid, metabolic products of Polyporus benzoinus (Wahl.) Fr. | 1952 | Biochem. J. | pmid:14925127 |
| CLAYTON RB and BLOCH K | Biological synthesis of lanosterol and agnosterol. | 1956 | J. Biol. Chem. | pmid:13278338 |
| CLAYTON RB and BLOCH K | The biological conversion of lanosterol to cholesterol. | 1956 | J. Biol. Chem. | pmid:13278339 |
| BLOCH K et al. | Synthesis of lanosterol in vivo. | 1957 | J. Biol. Chem. | pmid:13398396 |
| BLOCH K et al. | Isolation of lanosterol from isocholesterol. | 1957 | J. Biol. Chem. | pmid:13398397 |
| STOKES WM | The separation of the compounds of the lanosterol group present in "isocholesterol" by chromatography of the p-iodobenzoates-I 131 and benzoates. | 1957 | Arch. Biochem. Biophys. | pmid:13425621 |
| OLSON JA et al. | On the demethylation of lanosterol to cholesterol. | 1957 | J. Biol. Chem. | pmid:13438883 |
| TCHEN TT and BLOCH K | On the conversion of squalene to lanosterol in vitro. | 1957 | J. Biol. Chem. | pmid:13438881 |
| GAUTSCHI F and BLOCH K | Synthesis of isomeric 4,4-dimethylcholestenols and identification of a lanosterol metabolite. | 1958 | J. Biol. Chem. | pmid:13610839 |
| KANDUTSCH AA and RUSSELL AE | Preputial gland tumor sterols. I. The occurrence of 24,25-dihydrolanosterol and a comparison with liver and the normal gland. | 1959 | J. Biol. Chem. | pmid:13673010 |
| KANDUTSCH AA and RUSSELL AE | Preputial gland tumor sterols. 3. A metabolic pathway from lanosterol to cholesterol. | 1960 | J. Biol. Chem. | pmid:14404284 |
| KRITCHEVSKY D and DEFENDI V | Deposition of tritium labelled sterols (cholesterol, sitosterol, lanosterol) in brain and other organs of the growing chicken. | 1962 Jul-Aug | J. Neurochem. | pmid:14459731 |
| AVIGAN J et al. | Studies of cholesterol biosynthesis. IV. Reduction of lanosterol to 24,25-dihydrolanosterol by rat liver homogenates. | 1963 | J. Biol. Chem. | pmid:13965665 |
| LINDBERG M et al. | Ketonic intermediates in the demethylation of lanosterol. | 1963 | J. Biol. Chem. | pmid:13930748 |
| GAYLOR JL | BIOSYNTHESIS OF SKIN STEROLS. VI. ENZYMATIC DEMETHYLATION OF LANOSTEROL AND LANOSTA-7,24-DIEN-3BETA-OL BY HOMOGENATES OF RAT LIVER. | 1964 | J. Biol. Chem. | pmid:14154451 |