| MeSH term | MeSH ID | Detail |
|---|---|---|
| Xanthomatosis | D014973 | 17 associated lipids |
| Leukemia, Hairy Cell | D007943 | 5 associated lipids |
| Carcinoma, Hepatocellular | D006528 | 140 associated lipids |
| Abetalipoproteinemia | D000012 | 7 associated lipids |
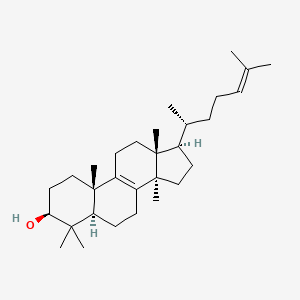
lanosterol
lanosterol is a lipid of Sterol Lipids (ST) class. Lanosterol is associated with abnormalities such as Infection, Atherosclerosis, Myocardial Infarction, Chagas Disease and Fatty Liver. The involved functions are known as Signal, Cytokinesis, physiological aspects, Stereochemistry and ergosterol biosynthetic process. Lanosterol often locates in Body tissue, Membrane, Plasma membrane, Tissue membrane and Cytoskeletal Filaments. The associated genes with lanosterol are Retinoic Acid Response Element, P4HTM gene, CYP51A1 gene, HM13 gene and SC4MOL gene. The related lipids are pneumocysterol, Sterols, lanosteryl acetate, ebericol and cycloartenol. The related experimental models are Knock-out.
Cross Reference
Introduction
To understand associated biological information of lanosterol, we collected biological information of abnormalities, associated pathways, cellular/molecular locations, biological functions, related genes/proteins, lipids and common seen animal/experimental models with organized paragraphs from literatures.
What diseases are associated with lanosterol?
lanosterol is suspected in Atherosclerosis, Chagas Disease, vaginalis, hypercholesterolemia, Obesity, Infection and other diseases in descending order of the highest number of associated sentences.
Related references are mostly published in these journals:
| Disease | Cross reference | Weighted score | Related literature |
|---|
Possible diseases from mapped MeSH terms on references
We collected disease MeSH terms mapped to the references associated with lanosterol
PubChem Associated disorders and diseases
What pathways are associated with lanosterol
Lipid pathways are not clear in current pathway databases. We organized associated pathways with lanosterol through full-text articles, including metabolic pathways or pathways of biological mechanisms.
Related references are published most in these journals:
| Pathway name | Related literatures |
|---|
PubChem Biomolecular Interactions and Pathways
Link to PubChem Biomolecular Interactions and PathwaysWhat cellular locations are associated with lanosterol?
Visualization in cellular structure
Associated locations are in red color. Not associated locations are in black.
Related references are published most in these journals:
| Location | Cross reference | Weighted score | Related literatures |
|---|
What functions are associated with lanosterol?
Related references are published most in these journals:
| Function | Cross reference | Weighted score | Related literatures |
|---|
What lipids are associated with lanosterol?
Related references are published most in these journals:
| Lipid concept | Cross reference | Weighted score | Related literatures |
|---|
What genes are associated with lanosterol?
Related references are published most in these journals:
| Gene | Cross reference | Weighted score | Related literatures |
|---|
What common seen animal models are associated with lanosterol?
Knock-out
Knock-out are used in the study 'Dual biosynthetic pathways to phytosterol via cycloartenol and lanosterol in Arabidopsis.' (Ohyama K et al., 2009), Knock-out are used in the study 'Expression, purification, and characterization of Aspergillus fumigatus sterol 14-alpha demethylase (CYP51) isoenzymes A and B.' (Warrilow AG et al., 2010) and Knock-out are used in the study 'Potential biological functions of cytochrome P450 reductase-dependent enzymes in small intestine: novel link to expression of major histocompatibility complex class II genes.' (D'Agostino J et al., 2012).
Related references are published most in these journals:
| Model | Cross reference | Weighted score | Related literatures |
|---|
NCBI Entrez Crosslinks
All references with lanosterol
Download all related citations| Authors | Title | Published | Journal | PubMed Link |
|---|---|---|---|---|
| Fan J et al. | Characterization of the sterol 14α-demethylases of Fusarium graminearum identifies a novel genus-specific CYP51 function. | 2013 | New Phytol. | pmid:23442154 |
| Madoui MA et al. | Sterol metabolism in the oomycete Aphanomyces euteiches, a legume root pathogen. | 2009 | New Phytol. | pmid:19496952 |
| Modica S et al. | Deciphering the nuclear bile acid receptor FXR paradigm. | 2010 | Nucl Recept Signal | pmid:21383957 |
| Sharman JL et al. | IUPHAR-DB: updated database content and new features. | 2013 | Nucleic Acids Res. | pmid:23087376 |
| Hu J et al. | Cellular cholesterol delivery, intracellular processing and utilization for biosynthesis of steroid hormones. | 2010 | Nutr Metab (Lond) | pmid:20515451 |
| Chung MJ et al. | Anticancer activity of subfractions containing pure compounds of Chaga mushroom (Inonotus obliquus) extract in human cancer cells and in Balbc/c mice bearing Sarcoma-180 cells. | 2010 | Nutr Res Pract | pmid:20607061 |
| Dai J et al. | Elimination of quiescent slow-cycling cells via reducing quiescence depth by natural compounds purified from Ganoderma lucidum. | 2017 | Oncotarget | pmid:28099150 |
| Skinner C and Miraldi Utz V | Pharmacological approaches to restoring lens transparency: Real world applications. | Ophthalmic Genet. | pmid:27648776 | |
| Matsuda SP et al. | Mechanistic insights into triterpene synthesis from quantum mechanical calculations. Detection of systematic errors in B3LYP cyclization energies. | 2006 | Org. Biomol. Chem. | pmid:16446812 |
| Winne JM et al. | Nonenzymic polycyclization of analogues of oxidosqualene with a preformed C-ring. | 2008 | Org. Biomol. Chem. | pmid:18480904 |
| Wu TK et al. | Mutation of isoleucine 705 of the oxidosqualene-lanosterol cyclase from Saccharomyces cerevisiae affects lanosterol's C/D-ring cyclization and 17α/β-exocyclic side chain stereochemistry. | 2011 | Org. Biomol. Chem. | pmid:21157613 |
| Yang XW et al. | Abiesatrines A-J: anti-inflammatory and antitumor triterpenoids from Abies georgei Orr. | 2010 | Org. Biomol. Chem. | pmid:20372737 |
| Hess BA | Formation of the C ring in the lanosterol biosynthesis from squalene. | 2003 | Org. Lett. | pmid:12529131 |
| Wu TK et al. | Importance of Saccharomyces cerevisiae oxidosqualene-lanosterol cyclase tyrosine 707 residue for chair-boat bicyclic ring formation and deprotonation reactions. | 2008 | Org. Lett. | pmid:18842050 |
| Wu TK et al. | Tryptophan 232 within oxidosqualene-lanosterol cyclase from Saccharomyces cerevisiae influences rearrangement and deprotonation but not cyclization reactions. | 2006 | Org. Lett. | pmid:16562881 |
| Meyer MM et al. | Directed evolution to generate cycloartenol synthase mutants that produce lanosterol. | 2002 | Org. Lett. | pmid:11950371 |
| Joubert BM et al. | Trypanosome and animal lanosterol synthases use different catalytic motifs. | 2001 | Org. Lett. | pmid:11405754 |
| Matsuda SP et al. | Steric bulk at cycloartenol synthase position 481 influences cyclization and deprotonation. | 2000 | Org. Lett. | pmid:10930258 |
| Hu ZX et al. | Kadcoccinones A-F, New Biogenetically Related Lanostane-Type Triterpenoids with Diverse Skeletons from Kadsura coccinea. | 2015 | Org. Lett. | pmid:26348535 |
| Liu YT et al. | Protein engineering of Saccharomyces cerevisiae oxidosqualene-lanosterol cyclase into parkeol synthase. | 2012 | Org. Lett. | pmid:23043506 |