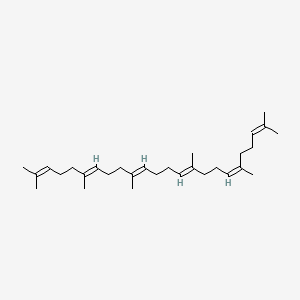
(e,e,e,e)-squalene
(e,e,e,e)-squalene is a lipid of Fatty Acyls (FA) class. (e,e,e,e)-squalene is associated with abnormalities such as Hypercholesterolemia and Cataract. The involved functions are known as Process, metaplastic cell transformation, Protein Overexpression, Anabolism and Biosynthetic Pathways. (e,e,e,e)-squalene often locates in Membrane, Protoplasm, Plasma membrane, Tissue membrane and Back. The associated genes with (e,e,e,e)-squalene are Genome, IMPACT gene, GAPDH gene, GTF2I gene and Chromatin. The related lipids are Membrane Lipids, cycloartenol, Sterols, Fatty Acids and Nonesterified Fatty Acids.
Cross Reference
Introduction
To understand associated biological information of (e,e,e,e)-squalene, we collected biological information of abnormalities, associated pathways, cellular/molecular locations, biological functions, related genes/proteins, lipids and common seen animal/experimental models with organized paragraphs from literatures.
What diseases are associated with (e,e,e,e)-squalene?
(e,e,e,e)-squalene is suspected in Hypercholesterolemia, Cataract and other diseases in descending order of the highest number of associated sentences.
Related references are mostly published in these journals:
| Disease | Cross reference | Weighted score | Related literature |
|---|
Possible diseases from mapped MeSH terms on references
We collected disease MeSH terms mapped to the references associated with (e,e,e,e)-squalene
PubChem Associated disorders and diseases
What pathways are associated with (e,e,e,e)-squalene
There are no associated biomedical information in the current reference collection.
PubChem Biomolecular Interactions and Pathways
Link to PubChem Biomolecular Interactions and PathwaysWhat cellular locations are associated with (e,e,e,e)-squalene?
Visualization in cellular structure
Associated locations are in red color. Not associated locations are in black.
Related references are published most in these journals:
| Location | Cross reference | Weighted score | Related literatures |
|---|
What functions are associated with (e,e,e,e)-squalene?
Related references are published most in these journals:
| Function | Cross reference | Weighted score | Related literatures |
|---|
What lipids are associated with (e,e,e,e)-squalene?
Related references are published most in these journals:
| Lipid concept | Cross reference | Weighted score | Related literatures |
|---|
What genes are associated with (e,e,e,e)-squalene?
Related references are published most in these journals:
| Gene | Cross reference | Weighted score | Related literatures |
|---|
What common seen animal models are associated with (e,e,e,e)-squalene?
There are no associated biomedical information in the current reference collection.
NCBI Entrez Crosslinks
All references with (e,e,e,e)-squalene
Download all related citations| Authors | Title | Published | Journal | PubMed Link |
|---|---|---|---|---|
| Minutello M et al. | Safety and immunogenicity of an inactivated subunit influenza virus vaccine combined with MF59 adjuvant emulsion in elderly subjects, immunized for three consecutive influenza seasons. | 1999 | Vaccine | pmid:9987141 |
| Madhun AS et al. | An adjuvanted pandemic influenza H1N1 vaccine provides early and long term protection in health care workers. | 2010 | Vaccine | pmid:21034828 |
| Esposito S et al. | Pandemic influenza A/H1N1 vaccine administered sequentially or simultaneously with seasonal influenza vaccine to HIV-infected children and adolescents. | 2011 | Vaccine | pmid:21199699 |
| Pellegrini M et al. | MF59-adjuvanted versus non-adjuvanted influenza vaccines: integrated analysis from a large safety database. | 2009 | Vaccine | pmid:19751689 |
| Forrest HL et al. | Single- and multiple-clade influenza A H5N1 vaccines induce cross protection in ferrets. | 2009 | Vaccine | pmid:19406182 |
| Camilloni B et al. | An influenza B outbreak during the 2007/2008 winter among appropriately immunized elderly people living in a nursing home. | 2010 | Vaccine | pmid:20846530 |
| Puig-Barberà J et al. | Effectiveness of MF59-adjuvanted subunit influenza vaccine in preventing hospitalisations for cardiovascular disease, cerebrovascular disease and pneumonia in the elderly. | 2007 | Vaccine | pmid:17889411 |
| Yu S et al. | Novel Th1-biased adjuvant, SPO1, enhances mucosal and systemic immunogenicity of vaccines administered intranasally in mice. | 2012 | Vaccine | pmid:22709954 |
| Chandramouli S et al. | Generation of a parvovirus B19 vaccine candidate. | 2013 | Vaccine | pmid:23827313 |
| Nielsen AB et al. | Immune response after one or two doses of pandemic influenza A (H1N1) monovalent, AS03-adjuvanted vaccine in HIV infected adults. | 2012 | Vaccine | pmid:23036498 |