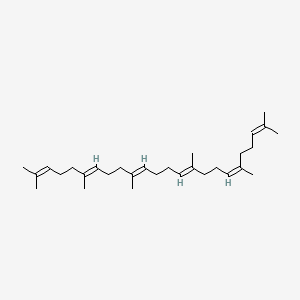
(e,e,e,e)-squalene
(e,e,e,e)-squalene is a lipid of Fatty Acyls (FA) class. (e,e,e,e)-squalene is associated with abnormalities such as Hypercholesterolemia and Cataract. The involved functions are known as Process, metaplastic cell transformation, Protein Overexpression, Anabolism and Biosynthetic Pathways. (e,e,e,e)-squalene often locates in Membrane, Protoplasm, Plasma membrane, Tissue membrane and Back. The associated genes with (e,e,e,e)-squalene are Genome, IMPACT gene, GAPDH gene, GTF2I gene and Chromatin. The related lipids are Membrane Lipids, cycloartenol, Sterols, Fatty Acids and Nonesterified Fatty Acids.
Cross Reference
Introduction
To understand associated biological information of (e,e,e,e)-squalene, we collected biological information of abnormalities, associated pathways, cellular/molecular locations, biological functions, related genes/proteins, lipids and common seen animal/experimental models with organized paragraphs from literatures.
What diseases are associated with (e,e,e,e)-squalene?
(e,e,e,e)-squalene is suspected in Hypercholesterolemia, Cataract and other diseases in descending order of the highest number of associated sentences.
Related references are mostly published in these journals:
| Disease | Cross reference | Weighted score | Related literature |
|---|
Possible diseases from mapped MeSH terms on references
We collected disease MeSH terms mapped to the references associated with (e,e,e,e)-squalene
PubChem Associated disorders and diseases
What pathways are associated with (e,e,e,e)-squalene
There are no associated biomedical information in the current reference collection.
PubChem Biomolecular Interactions and Pathways
Link to PubChem Biomolecular Interactions and PathwaysWhat cellular locations are associated with (e,e,e,e)-squalene?
Visualization in cellular structure
Associated locations are in red color. Not associated locations are in black.
Related references are published most in these journals:
| Location | Cross reference | Weighted score | Related literatures |
|---|
What functions are associated with (e,e,e,e)-squalene?
Related references are published most in these journals:
| Function | Cross reference | Weighted score | Related literatures |
|---|
What lipids are associated with (e,e,e,e)-squalene?
Related references are published most in these journals:
| Lipid concept | Cross reference | Weighted score | Related literatures |
|---|
What genes are associated with (e,e,e,e)-squalene?
Related references are published most in these journals:
| Gene | Cross reference | Weighted score | Related literatures |
|---|
What common seen animal models are associated with (e,e,e,e)-squalene?
There are no associated biomedical information in the current reference collection.
NCBI Entrez Crosslinks
All references with (e,e,e,e)-squalene
Download all related citations| Authors | Title | Published | Journal | PubMed Link |
|---|---|---|---|---|
| Michael-Jubeli R et al. | High-temperature gas chromatography-mass spectrometry for skin surface lipids profiling. | 2011 | J. Lipid Res. | pmid:20952798 |
| Lama-Muñoz A et al. | New hydrothermal treatment of alperujo enhances the content of bioactive minor components in crude pomace olive oil. | 2011 | J. Agric. Food Chem. | pmid:21265536 |
| Ellebedy AH et al. | Inflammasome-independent role of the apoptosis-associated speck-like protein containing CARD (ASC) in the adjuvant effect of MF59. | 2011 | Proc. Natl. Acad. Sci. U.S.A. | pmid:21270336 |
| Morel S et al. | Adjuvant System AS03 containing α-tocopherol modulates innate immune response and leads to improved adaptive immunity. | 2011 | Vaccine | pmid:21256188 |
| Sarpietro MG et al. | Synthesis of n-squalenoyl cytarabine and evaluation of its affinity with phospholipid bilayers and monolayers. | 2011 | Int J Pharm | pmid:21219999 |
| Fenoglio D et al. | MF-59 adjuvant influence on the functions of gammadelta T cells in HIV-1+ adults immunized with influenza seasonal vaccine. | 2011 | J Prev Med Hyg | pmid:22010544 |
| Precioso AR et al. | A phase I randomized, double-blind, controlled trial of 2009 influenza A (H1N1) inactivated monovalent vaccines with different adjuvant systems. | 2011 | Vaccine | pmid:21945258 |
| Pariani E et al. | Response to 2009 pandemic and seasonal influenza vaccines co-administered to HIV-infected and HIV-uninfected former drug users living in a rehabilitation community in Italy. | 2011 | Vaccine | pmid:21974995 |
| Fox CB et al. | Effects of emulsifier concentration, composition, and order of addition in squalene-phosphatidylcholine oil-in-water emulsions. | 2011 | Pharm Dev Technol | pmid:20550484 |
| Pollard AJ and Hill AV | Antibody repertoire: embracing diversity. | 2011 | Sci Transl Med | pmid:21795586 |
| Okike IO et al. | The immunogenicity of a novel A (H1N1) vaccine in HIV-infected children. | 2011 | Vaccine | pmid:21742005 |
| Kowert BA and Watson MB | Diffusion of organic solutes in squalane. | 2011 | J Phys Chem B | pmid:21780767 |
| Mackay D and Jones PJ | Evaluation of methods for the determination of cholesterol absorption and synthesis in humans. | 2011 | Atherosclerosis | pmid:21763652 |
| Jawaid AM et al. | Shape-controlled colloidal synthesis of rock-salt lead selenide nanocrystals. | 2011 | ACS Nano | pmid:21770427 |
| Oliaro-Bosso S et al. | Characterization of the channel constriction allowing the access of the substrate to the active site of yeast oxidosqualene cyclase. | 2011 | PLoS ONE | pmid:21811565 |
| Bildstein L et al. | Interaction of an amphiphilic squalenoyl prodrug of gemcitabine with cellular membranes. | 2011 | Eur J Pharm Biopharm | pmid:21784150 |
| Iannazzo S | Pharmacoeconomic evaluation of the MF59--adjuvanted influenza vaccine in the elderly population in Italy. | 2011 | J Prev Med Hyg | pmid:21710816 |
| Seubert A et al. | Adjuvanticity of the oil-in-water emulsion MF59 is independent of Nlrp3 inflammasome but requires the adaptor protein MyD88. | 2011 | Proc. Natl. Acad. Sci. U.S.A. | pmid:21690334 |
| Khurana S et al. | MF59 adjuvant enhances diversity and affinity of antibody-mediated immune response to pandemic influenza vaccines. | 2011 | Sci Transl Med | pmid:21632986 |
| Zhang LW et al. | Oil components modulate the skin delivery of 5-aminolevulinic acid and its ester prodrug from oil-in-water and water-in-oil nanoemulsions. | 2011 | Int J Nanomedicine | pmid:21556344 |
| Lens M and Podesta Marty MH | Assessment of the kinetics of the antioxidative capacity of topical antioxidants. | 2011 | J Drugs Dermatol | pmid:21369642 |
| Cristiani C et al. | Safety of MF-59 adjuvanted vaccine for pandemic influenza: results of the vaccination campaign in an Italian health district. | 2011 | Vaccine | pmid:21396903 |
| Tabache F et al. | Acute polyarthritis after influenza A (H1N1) immunization. | 2011 | Joint Bone Spine | pmid:21444232 |
| Sun Y et al. | A reproducible in-vivo model of lymphatic malformation in rats. | 2011 | J. Comp. Pathol. | pmid:21419420 |
| Montana M et al. | Safety review: squalene and thimerosal in vaccines. | 2010 Nov-Dec | Therapie | pmid:21176760 |
| Voyron S et al. | Antifungal activity of bis-azasqualenes, inhibitors of oxidosqualene cyclase. | 2010 | Mycoses | pmid:19549106 |
| Brunner R et al. | The ABC of clinical and experimental adjuvants--a brief overview. | 2010 | Immunol. Lett. | pmid:19895847 |
| Eroglu E and Melis A | Extracellular terpenoid hydrocarbon extraction and quantitation from the green microalgae Botryococcus braunii var. Showa. | 2010 | Bioresour. Technol. | pmid:20005092 |
| Kalvodova L | Squalene-based oil-in-water emulsion adjuvants perturb metabolism of neutral lipids and enhance lipid droplet formation. | 2010 | Biochem. Biophys. Res. Commun. | pmid:20018176 |
| Kimura M et al. | Protostadienol synthase from Aspergillus fumigatus: functional conversion into lanosterol synthase. | 2010 | Biochem. Biophys. Res. Commun. | pmid:19951700 |
| Ansaldi F et al. | Antibody response against heterogeneous circulating influenza virus strains elicited by MF59- and non-adjuvanted vaccines during seasons with good or partial matching between vaccine strain and clinical isolates. | 2010 | Vaccine | pmid:20433807 |
| Sugihara N et al. | Novel fractionation method for squalene and phytosterols contained in the deodorization distillate of rice bran oil. | 2010 | J Oleo Sci | pmid:20103978 |
| Chen CC et al. | Effects of lipophilic emulsifiers on the oral administration of lovastatin from nanostructured lipid carriers: physicochemical characterization and pharmacokinetics. | 2010 | Eur J Pharm Biopharm | pmid:20060469 |
| Spanova M et al. | Effect of lipid particle biogenesis on the subcellular distribution of squalene in the yeast Saccharomyces cerevisiae. | 2010 | J. Biol. Chem. | pmid:20032462 |
| Tsai T et al. | Exposure to MF59-adjuvanted influenza vaccines during pregnancy--a retrospective analysis. | 2010 | Vaccine | pmid:19969117 |
| Pietrella D et al. | A beta-glucan-conjugate vaccine and anti-beta-glucan antibodies are effective against murine vaginal candidiasis as assessed by a novel in vivo imaging technique. | 2010 | Vaccine | pmid:20038431 |
| Gasparini R et al. | Impact of prior or concomitant seasonal influenza vaccination on MF59-adjuvanted H1N1v vaccine (Focetria) in adult and elderly subjects. | 2010 | Int. J. Clin. Pract. | pmid:20039974 |
| De Luca C and Valacchi G | Surface lipids as multifunctional mediators of skin responses to environmental stimuli. | 2010 | Mediators Inflamm. | pmid:20981292 |
| Tsuchiya M et al. | Cholesterol biosynthesis pathway intermediates and inhibitors regulate glucose-stimulated insulin secretion and secretory granule formation in pancreatic beta-cells. | 2010 | Endocrinology | pmid:20685866 |
| Camilloni B et al. | An influenza B outbreak during the 2007/2008 winter among appropriately immunized elderly people living in a nursing home. | 2010 | Vaccine | pmid:20846530 |
| Bildstein L et al. | Transmembrane diffusion of gemcitabine by a nanoparticulate squalenoyl prodrug: an original drug delivery pathway. | 2010 | J Control Release | pmid:20691740 |
| Weiss TL et al. | Raman spectroscopy analysis of botryococcene hydrocarbons from the green microalga Botryococcus braunii. | 2010 | J. Biol. Chem. | pmid:20705610 |
| Nakagawa K et al. | gamma-Tocotrienol reduces squalene hydroperoxide-induced inflammatory responses in HaCaT keratinocytes. | 2010 | Lipids | pmid:20714817 |
| Black S et al. | Safety of MF59-adjuvanted versus non-adjuvanted influenza vaccines in children and adolescents: an integrated analysis. | 2010 | Vaccine | pmid:20813217 |
| Vesikari T et al. | Immunogenicity and safety of MF59-adjuvanted H5N1 influenza vaccine from infancy to adolescence. | 2010 | Pediatrics | pmid:20819892 |
| Pili B et al. | Liposomal squalenoyl-gemcitabine: formulation, characterization and anticancer activity evaluation. | 2010 | Nanoscale | pmid:20820745 |
| Camera E et al. | Comprehensive analysis of the major lipid classes in sebum by rapid resolution high-performance liquid chromatography and electrospray mass spectrometry. | 2010 | J. Lipid Res. | pmid:20719760 |
| Watanabe T et al. | Novel pyrrole- and 1,2,3-triazole-based 2,3-oxidosqualene cyclase inhibitors. | 2010 | Bioorg. Med. Chem. Lett. | pmid:20728352 |
| Ray R et al. | Characterization of antibodies induced by vaccination with hepatitis C virus envelope glycoproteins. | 2010 | J. Infect. Dis. | pmid:20677942 |
| Beran J et al. | A phase III, randomized, open-label study to assess the tolerability and immunogenicity of an H5N1 influenza vaccine administered to healthy adults with a 1-, 2-, 3-, or 6-week interval between first and second doses. | 2010 | Clin Ther | pmid:21316535 |