| MeSH term | MeSH ID | Detail |
|---|---|---|
| Leukemia P388 | D007941 | 43 associated lipids |
| Leukemia, Experimental | D007942 | 42 associated lipids |
| Leukemia, Hairy Cell | D007943 | 5 associated lipids |
| Lipodystrophy | D008060 | 4 associated lipids |
| Liver Cirrhosis | D008103 | 67 associated lipids |
| Lung Neoplasms | D008175 | 171 associated lipids |
| Lupus Erythematosus, Systemic | D008180 | 43 associated lipids |
| Metabolism, Inborn Errors | D008661 | 46 associated lipids |
| Nervous System Diseases | D009422 | 37 associated lipids |
| Neuroblastoma | D009447 | 66 associated lipids |
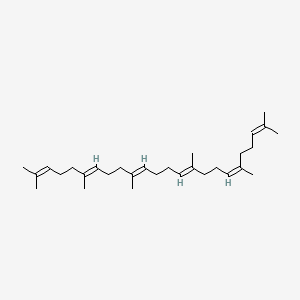
(e,e,e,e)-squalene
(e,e,e,e)-squalene is a lipid of Fatty Acyls (FA) class. (e,e,e,e)-squalene is associated with abnormalities such as Hypercholesterolemia and Cataract. The involved functions are known as Process, metaplastic cell transformation, Protein Overexpression, Anabolism and Biosynthetic Pathways. (e,e,e,e)-squalene often locates in Membrane, Protoplasm, Plasma membrane, Tissue membrane and Back. The associated genes with (e,e,e,e)-squalene are Genome, IMPACT gene, GAPDH gene, GTF2I gene and Chromatin. The related lipids are Membrane Lipids, cycloartenol, Sterols, Fatty Acids and Nonesterified Fatty Acids.
Cross Reference
Introduction
To understand associated biological information of (e,e,e,e)-squalene, we collected biological information of abnormalities, associated pathways, cellular/molecular locations, biological functions, related genes/proteins, lipids and common seen animal/experimental models with organized paragraphs from literatures.
What diseases are associated with (e,e,e,e)-squalene?
(e,e,e,e)-squalene is suspected in Hypercholesterolemia, Cataract and other diseases in descending order of the highest number of associated sentences.
Related references are mostly published in these journals:
| Disease | Cross reference | Weighted score | Related literature |
|---|
Possible diseases from mapped MeSH terms on references
We collected disease MeSH terms mapped to the references associated with (e,e,e,e)-squalene
PubChem Associated disorders and diseases
What pathways are associated with (e,e,e,e)-squalene
There are no associated biomedical information in the current reference collection.
PubChem Biomolecular Interactions and Pathways
Link to PubChem Biomolecular Interactions and PathwaysWhat cellular locations are associated with (e,e,e,e)-squalene?
Visualization in cellular structure
Associated locations are in red color. Not associated locations are in black.
Related references are published most in these journals:
| Location | Cross reference | Weighted score | Related literatures |
|---|
What functions are associated with (e,e,e,e)-squalene?
Related references are published most in these journals:
| Function | Cross reference | Weighted score | Related literatures |
|---|
What lipids are associated with (e,e,e,e)-squalene?
Related references are published most in these journals:
| Lipid concept | Cross reference | Weighted score | Related literatures |
|---|
What genes are associated with (e,e,e,e)-squalene?
Related references are published most in these journals:
| Gene | Cross reference | Weighted score | Related literatures |
|---|
What common seen animal models are associated with (e,e,e,e)-squalene?
There are no associated biomedical information in the current reference collection.
NCBI Entrez Crosslinks
All references with (e,e,e,e)-squalene
Download all related citations| Authors | Title | Published | Journal | PubMed Link |
|---|---|---|---|---|
| Byars NE and Allison AC | Adjuvant formulation for use in vaccines to elicit both cell-mediated and humoral immunity. | 1987 | Vaccine | pmid:3499713 |
| Cataldo DM and Van Nest G | The adjuvant MF59 increases the immunogenicity and protective efficacy of subunit influenza vaccine in mice. | 1997 | Vaccine | pmid:9364672 |
| Barchfeld GL et al. | The adjuvants MF59 and LT-K63 enhance the mucosal and systemic immunogenicity of subunit influenza vaccine administered intranasally in mice. | 1999 | Vaccine | pmid:10067675 |
| Dupuis M et al. | Distribution of adjuvant MF59 and antigen gD2 after intramuscular injection in mice. | 1999 | Vaccine | pmid:10519932 |
| Podda A | The adjuvanted influenza vaccines with novel adjuvants: experience with the MF59-adjuvanted vaccine. | 2001 | Vaccine | pmid:11257408 |
| Greer CE et al. | The comparison of the effect of LTR72 and MF59 adjuvants on mouse humoral response to intranasal immunisation with human papillomavirus type 6b (HPV-6b) virus-like particles. | 2000 | Vaccine | pmid:11137233 |
| Skeiky YA et al. | Protective efficacy of a tandemly linked, multi-subunit recombinant leishmanial vaccine (Leish-111f) formulated in MPL adjuvant. | 2002 | Vaccine | pmid:12213399 |
| Stephenson I et al. | Boosting immunity to influenza H5N1 with MF59-adjuvanted H5N3 A/Duck/Singapore/97 vaccine in a primed human population. | 2003 | Vaccine | pmid:12639491 |
| Romera SA et al. | Adjuvant effects of sulfolipo-cyclodextrin in a squalane-in-water and water-in-mineral oil emulsions for BHV-1 vaccines in cattle. | 2000 | Vaccine | pmid:10924795 |
| Boyce TG et al. | Safety and immunogenicity of adjuvanted and unadjuvanted subunit influenza vaccines administered intranasally to healthy adults. | 2000 | Vaccine | pmid:10930676 |
| O'Hagan DT et al. | Microparticles in MF59, a potent adjuvant combination for a recombinant protein vaccine against HIV-1. | 2000 | Vaccine | pmid:10699327 |
| Süli J et al. | Experimental squalene adjuvant. I. Preparation and testing of its effectiveness. | 2004 | Vaccine | pmid:15308373 |
| BenÃsek Z et al. | Experimental squalene adjuvant. II. Harmlessness and local reactogenity. | 2004 | Vaccine | pmid:15308374 |
| Cristiani C et al. | Safety of MF-59 adjuvanted vaccine for pandemic influenza: results of the vaccination campaign in an Italian health district. | 2011 | Vaccine | pmid:21396903 |
| Meier S et al. | Antibody responses to natural influenza A/H1N1/09 disease or following immunization with adjuvanted vaccines, in immunocompetent and immunocompromised children. | 2011 | Vaccine | pmid:21419775 |
| Morel S et al. | Adjuvant System AS03 containing α-tocopherol modulates innate immune response and leads to improved adaptive immunity. | 2011 | Vaccine | pmid:21256188 |
| Tritto E et al. | Mechanism of action of licensed vaccine adjuvants. | 2009 | Vaccine | pmid:19200813 |
| Ansaldi F et al. | Adjuvanted seasonal influenza vaccines and perpetual viral metamorphosis: the importance of cross-protection. | 2009 | Vaccine | pmid:19200846 |
| Phillips CJ et al. | Antibodies to squalene in US Navy Persian Gulf War veterans with chronic multisymptom illness. | 2009 | Vaccine | pmid:19379786 |
| Keitel W et al. | Dose ranging of adjuvant and antigen in a cell culture H5N1 influenza vaccine: safety and immunogenicity of a phase 1/2 clinical trial. | 2010 | Vaccine | pmid:19835829 |
| Vesikari T et al. | MF59-adjuvanted influenza vaccine (FLUAD) in children: safety and immunogenicity following a second year seasonal vaccination. | 2009 | Vaccine | pmid:19840662 |
| Frey SE et al. | Safety and immunogenicity of HCV E1E2 vaccine adjuvanted with MF59 administered to healthy adults. | 2010 | Vaccine | pmid:20619382 |
| Dell'Era L et al. | Immunogenicity, safety and tolerability of MF59-adjuvanted seasonal influenza vaccine in children with juvenile idiopathic arthritis. | 2012 | Vaccine | pmid:22138210 |
| Okike IO et al. | The immunogenicity of a novel A (H1N1) vaccine in HIV-infected children. | 2011 | Vaccine | pmid:21742005 |
| Della Cioppa G et al. | Trivalent and quadrivalent MF59(®)-adjuvanted influenza vaccine in young children: a dose- and schedule-finding study. | 2011 | Vaccine | pmid:21906647 |
| Fox CB et al. | Immunomodulatory and physical effects of oil composition in vaccine adjuvant emulsions. | 2011 | Vaccine | pmid:21906648 |
| Guillon C et al. | Formulation of HIV-1 Tat and p24 antigens by PLA nanoparticles or MF59 impacts the breadth, but not the magnitude, of serum and faecal antibody responses in rabbits. | 2007 | Vaccine | pmid:17904700 |
| Baldo V et al. | MF59-adjuvanted influenza vaccine confers superior immunogenicity in adult subjects (18-60 years of age) with chronic diseases who are at risk of post-influenza complications. | 2007 | Vaccine | pmid:17383057 |
| Bråve A et al. | Candidate HIV-1 gp140DeltaV2, Gag and Tat vaccines protect against experimental HIV-1/MuLV challenge. | 2007 | Vaccine | pmid:17707956 |
| Ansaldi F et al. | Cross-protection by MF59-adjuvanted influenza vaccine: neutralizing and haemagglutination-inhibiting antibody activity against A(H3N2) drifted influenza viruses. | 2008 | Vaccine | pmid:18294741 |
| de Roux A et al. | Impact of corticosteroids on the immune response to a MF59-adjuvanted influenza vaccine in elderly COPD-patients. | 2006 | Vaccine | pmid:16288937 |
| Singh M et al. | A preliminary evaluation of alternative adjuvants to alum using a range of established and new generation vaccine antigens. | 2006 | Vaccine | pmid:16300864 |
| Del Giudice G et al. | An MF59-adjuvanted inactivated influenza vaccine containing A/Panama/1999 (H3N2) induced broader serological protection against heterovariant influenza virus strain A/Fujian/2002 than a subunit and a split influenza vaccine. | 2006 | Vaccine | pmid:16464520 |
| Faenzi E et al. | One dose of an MF59-adjuvanted pandemic A/H1N1 vaccine recruits pre-existing immune memory and induces the rapid rise of neutralizing antibodies. | 2012 | Vaccine | pmid:22521851 |
| Hatz C et al. | A randomised, single-blind, dose-range study to assess the immunogenicity and safety of a cell-culture-derived A/H1N1 influenza vaccine in adult and elderly populations. | 2012 | Vaccine | pmid:22626675 |
| Precioso AR et al. | A phase I randomized, double-blind, controlled trial of 2009 influenza A (H1N1) inactivated monovalent vaccines with different adjuvant systems. | 2011 | Vaccine | pmid:21945258 |
| Pariani E et al. | Response to 2009 pandemic and seasonal influenza vaccines co-administered to HIV-infected and HIV-uninfected former drug users living in a rehabilitation community in Italy. | 2011 | Vaccine | pmid:21974995 |
| Andrews NJ et al. | Predictors of immune response and reactogenicity to AS03B-adjuvanted split virion and non-adjuvanted whole virion H1N1 (2009) pandemic influenza vaccines. | 2011 | Vaccine | pmid:21875635 |
| Candela S et al. | An early (3-6 weeks) active surveillance study to assess the safety of pandemic influenza vaccine Focetria in a province of Emilia-Romagna region, Italy - part one. | 2013 | Vaccine | pmid:22766247 |
| Langley JM et al. | Safety and immunogenicity of 2010-2011 H1N12009-containing trivalent inactivated influenza vaccine in children 12-59 months of age previously given AS03-adjuvanted H1N12009 pandemic vaccine: a PHAC/CIHR Influenza Research Network (PCIRN) study. | 2012 | Vaccine | pmid:22469860 |
| Fukase H et al. | Assessment of the immunogenicity and safety of varying doses of an MF59®-adjuvanted cell culture-derived A/H1N1 pandemic influenza vaccine in Japanese paediatric, adult and elderly subjects. | 2012 | Vaccine | pmid:22472791 |
| Hatz C et al. | A randomized clinical trial to identify the optimal antigen and MF59(®) adjuvant dose of a monovalent A/H1N1 pandemic influenza vaccine in healthy adult and elderly subjects. | 2012 | Vaccine | pmid:22446638 |
| Reynales H et al. | A prospective observational safety study on MF59(®) adjuvanted cell culture-derived vaccine, Celtura(®) during the A/H1N1 (2009) influenza pandemic. | 2012 | Vaccine | pmid:22902681 |
| Ventura R et al. | Technology transfer of an oil-in-water vaccine-adjuvant for strengthening pandemic influenza preparedness in Indonesia. | 2013 | Vaccine | pmid:22884665 |
| Moro ML et al. | A population based cohort study to assess the safety of pandemic influenza vaccine Focetria in Emilia-Romagna region, Italy - part two. | 2013 | Vaccine | pmid:22885015 |
| Calabro S et al. | The adjuvant effect of MF59 is due to the oil-in-water emulsion formulation, none of the individual components induce a comparable adjuvant effect. | 2013 | Vaccine | pmid:23684834 |
| Segura-Velázquez R et al. | Influenza vaccine: development of a novel intranasal and subcutaneous recombinant adjuvant. | 2013 | Vaccine | pmid:23746458 |
| Puig-Barberà J et al. | MF59-adjuvanted and virosomal influenza vaccines for preventing influenza hospitalization in older people: comparative effectiveness using the Valencia health care information system. | 2013 | Vaccine | pmid:23731629 |
| Van Buynder PG et al. | The comparative effectiveness of adjuvanted and unadjuvanted trivalent inactivated influenza vaccine (TIV) in the elderly. | 2013 | Vaccine | pmid:23933368 |
| Vinay TN et al. | Inactivated vaccine against viral hemorrhagic septicemia (VHS) emulsified with squalene and aluminum hydroxide adjuvant provides long term protection in olive flounder (Paralichthys olivaceus). | 2013 | Vaccine | pmid:23896420 |