| MeSH term | MeSH ID | Detail |
|---|---|---|
| Burns | D002056 | 34 associated lipids |
| Body Weight | D001835 | 333 associated lipids |
| Edema | D004487 | 152 associated lipids |
| Arthritis | D001168 | 41 associated lipids |
| Abscess | D000038 | 13 associated lipids |
| Psoriasis | D011565 | 47 associated lipids |
| Hypertension, Renovascular | D006978 | 10 associated lipids |
| Blood Loss, Surgical | D016063 | 6 associated lipids |
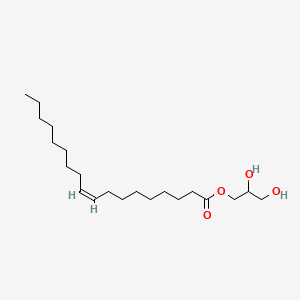
2,3-Dihydroxypropyl oleate
2,3-Dihydroxypropyl oleate is a lipid of Glycerolipids (GL) class. The involved functions are known as enzyme activity and acyltransferase activity. 2,3-dihydroxypropyl oleate often locates in soluble fraction.
Cross Reference
Introduction
To understand associated biological information of 2,3-Dihydroxypropyl oleate, we collected biological information of abnormalities, associated pathways, cellular/molecular locations, biological functions, related genes/proteins, lipids and common seen animal/experimental models with organized paragraphs from literatures.
What diseases are associated with 2,3-Dihydroxypropyl oleate?
There are no associated biomedical information in the current reference collection.
Possible diseases from mapped MeSH terms on references
We collected disease MeSH terms mapped to the references associated with 2,3-Dihydroxypropyl oleate
PubChem Associated disorders and diseases
What pathways are associated with 2,3-Dihydroxypropyl oleate
There are no associated biomedical information in the current reference collection.
PubChem Biomolecular Interactions and Pathways
Link to PubChem Biomolecular Interactions and PathwaysWhat cellular locations are associated with 2,3-Dihydroxypropyl oleate?
Visualization in cellular structure
Associated locations are in red color. Not associated locations are in black.
Related references are published most in these journals:
| Location | Cross reference | Weighted score | Related literatures |
|---|
What functions are associated with 2,3-Dihydroxypropyl oleate?
Related references are published most in these journals:
| Function | Cross reference | Weighted score | Related literatures |
|---|
What lipids are associated with 2,3-Dihydroxypropyl oleate?
There are no associated biomedical information in the current reference collection.
What genes are associated with 2,3-Dihydroxypropyl oleate?
There are no associated biomedical information in the current reference collection.
What common seen animal models are associated with 2,3-Dihydroxypropyl oleate?
There are no associated biomedical information in the current reference collection.
NCBI Entrez Crosslinks
All references with 2,3-Dihydroxypropyl oleate
Download all related citations| Authors | Title | Published | Journal | PubMed Link |
|---|---|---|---|---|
| Quiñones OG et al. | In vitro and in vivo influence of penetration enhancers in the topical application of celecoxib. | 2014 | Drug Dev Ind Pharm | pmid:23826859 |
| Silva JP et al. | Structural dynamics and physicochemical properties of pDNA/DODAB:MO lipoplexes: effect of pH and anionic lipids in inverted non-lamellar phases versus lamellar phases. | 2014 | Biochim. Biophys. Acta | pmid:24976292 |
| Zhou Y et al. | Biological artificial fluid-induced non-lamellar phases in glyceryl monooleate: the kinetics pathway and its digestive process by bile salts. | 2014 | Drug Dev Ind Pharm | pmid:23350691 |
| Madheswaran T et al. | Enhanced topical delivery of finasteride using glyceryl monooleate-based liquid crystalline nanoparticles stabilized by cremophor surfactants. | 2014 | AAPS PharmSciTech | pmid:24222268 |
| Morsi NM et al. | Silver sulfadiazine based cubosome hydrogels for topical treatment of burns: development and in vitro/in vivo characterization. | 2014 | Eur J Pharm Biopharm | pmid:23688805 |
| Esposito E et al. | Effect of nanostructured lipid vehicles on percutaneous absorption of curcumin. | 2014 | Eur J Pharm Biopharm | pmid:24361485 |
| Li Y et al. | Improving oral bioavailability of metformin hydrochloride using water-in-oil microemulsions and analysis of phase behavior after dilution. | 2014 | Int J Pharm | pmid:25014370 |
| Oka T and Hojo H | Single crystallization of an inverse bicontinuous cubic phase of a lipid. | 2014 | Langmuir | pmid:25007349 |
| Bye N et al. | Nitroxide-loaded hexosomes provide MRI contrast in vivo. | 2014 | Langmuir | pmid:24979524 |
| Oka T et al. | Initial step of pH-jump-induced lamellar to bicontinuous cubic phase transition in dioleoylphosphatidylserine/monoolein. | 2014 | Langmuir | pmid:24949525 |
| Wang L et al. | In vitro and in vivo evaluation of chitosan graft glyceryl monooleate as peroral delivery carrier of enoxaparin. | 2014 | Int J Pharm | pmid:24882036 |
| Du LR et al. | Development and evaluation of liquid embolic agents based on liquid crystalline material of glyceryl monooleate. | 2014 | Int J Pharm | pmid:24858389 |
| Maselko J et al. | Emergence of complex behavior in chemical cells: the system AlCl₃-NaOH. | 2014 | Langmuir | pmid:24805150 |
| Cohen-Avrahami M et al. | HIV-TAT enhances the transdermal delivery of NSAID drugs from liquid crystalline mesophases. | 2014 | J Phys Chem B | pmid:24798650 |
| Kolev VL et al. | Unit cell structure of water-filled monoolein into inverted hexagonal (H(II)) mesophase modeled by molecular dynamics. | 2014 | J Phys Chem B | pmid:24787641 |
| Ghattas N et al. | Monoolein production by triglycerides hydrolysis using immobilized Rhizopus oryzae lipase. | 2014 | Int. J. Biol. Macromol. | pmid:24755261 |
| Oliveira AC et al. | Dioctadecyldimethylammonium:monoolein nanocarriers for efficient in vitro gene silencing. | 2014 | ACS Appl Mater Interfaces | pmid:24712543 |
| Bitan-Cherbakovsky L et al. | Reversed hexagonal lyotropic liquid-crystal and open-shell glycodendrimers as potential vehicles for sustained release of sodium diclofenac. | 2014 | J Phys Chem B | pmid:24617448 |
| Tiossi RF et al. | In vitro and in vivo evaluation of the delivery of topical formulations containing glycoalkaloids of Solanum lycocarpum fruits. | 2014 | Eur J Pharm Biopharm | pmid:24509413 |
| Mengesha AE et al. | Binary blend of glyceryl monooleate and glyceryl monostearate for magnetically induced thermo-responsive local drug delivery system. | 2013 | Pharm. Res. | pmid:24158728 |
| Mishraki T et al. | Modulation of physical properties of reverse hexagonal mesophases: a dielectric spectroscopy study. | 2013 | J Colloid Interface Sci | pmid:23415506 |
| Réeff J et al. | Characterization and optimization of GMO-based gels with long term release for intraarticular administration. | 2013 | Int J Pharm | pmid:23651644 |
| Puglia C et al. | Evaluation of monooleine aqueous dispersions as tools for topical administration of curcumin: characterization, in vitro and ex-vivo studies. | 2013 | J Pharm Sci | pmid:23686742 |
| Patil SS et al. | Mapping ion-induced mesophasic transformation in lyotropic in situ gelling system and its correlation with pharmaceutical performance. | 2013 | Pharm. Res. | pmid:23595880 |
| Murgia S et al. | Drug-loaded fluorescent cubosomes: versatile nanoparticles for potential theranostic applications. | 2013 | Langmuir | pmid:23642193 |
| Squires AM et al. | Preparation of films of a highly aligned lipid cubic phase. | 2013 | Langmuir | pmid:23347289 |
| Bilalov A et al. | Embedding DNA in surfactant mesophases: the phase diagram of the ternary system dodecyltrimethylammonium-DNA/monoolein/water in comparison to the DNA-free analogue. | 2013 | J Colloid Interface Sci | pmid:23347998 |
| Hosmer JM et al. | Lamellar liquid crystalline phases for cutaneous delivery of Paclitaxel: impact of the monoglyceride. | 2013 | Pharm. Res. | pmid:23135821 |
| Réeff J et al. | New sustained-release intraarticular gel formulations based on monolein for local treatment of arthritic diseases. | 2013 | Drug Dev Ind Pharm | pmid:23078519 |
| Sato H et al. | Development of cyclosporine A-loaded dry-emulsion formulation using highly purified glycerol monooleate for safe inhalation therapy. | 2013 | Int J Pharm | pmid:23528280 |
| Evenbratt H et al. | In vivo study of an instantly formed lipid-water cubic phase formulation for efficient topical delivery of aminolevulinic acid and methyl-aminolevulinate. | 2013 | Int J Pharm | pmid:23727140 |
| Vicentini FT et al. | Liquid crystalline phase nanodispersions enable skin delivery of siRNA. | 2013 | Eur J Pharm Biopharm | pmid:23010565 |
| Li J et al. | A potential carrier based on liquid crystal nanoparticles for ophthalmic delivery of pilocarpine nitrate. | 2013 | Int J Pharm | pmid:23916822 |
| Bitan-Cherbakovsky L et al. | Structural characterization of lyotropic liquid crystals containing a dendrimer for solubilization and release of gallic acid. | 2013 | Colloids Surf B Biointerfaces | pmid:23973908 |
| Liu H et al. | The effect of surface charge of glycerol monooleate-based nanoparticles on the round window membrane permeability and cochlear distribution. | 2013 | J Drug Target | pmid:23944216 |
| Dai J and Kim JC | Photo responsive monoolein cubic phase containing coumarin-Tween 20 conjugates. | 2013 | Drug Dev Ind Pharm | pmid:23902365 |
| Tangso KJ et al. | Novel spiropyran amphiphiles and their application as light-responsive liquid crystalline components. | 2013 | J Phys Chem B | pmid:23909814 |
| Esposito E et al. | Curcumin containing monoolein aqueous dispersions: a preformulative study. | 2013 | Mater Sci Eng C Mater Biol Appl | pmid:24094206 |
| Khvostichenko DS et al. | Effects of detergent β-octylglucoside and phosphate salt solutions on phase behavior of monoolein mesophases. | 2013 | Biophys. J. | pmid:24138861 |
| Zulim Botega DC et al. | Development of formulations and processes to incorporate wax oleogels in ice cream. | 2013 | J. Food Sci. | pmid:24329951 |
| Saadeh M et al. | Reactive oxygen species stimulate insulin secretion in rat pancreatic islets: studies using mono-oleoyl-glycerol. | 2012 | PLoS ONE | pmid:22272304 |
| Panapisal V et al. | Formulation of microemulsion systems for dermal delivery of silymarin. | 2012 | AAPS PharmSciTech | pmid:22350738 |
| Kubicek J et al. | Controlled in meso phase crystallization--a method for the structural investigation of membrane proteins. | 2012 | PLoS ONE | pmid:22536388 |
| Moebus K et al. | Cubic phase-forming dry powders for controlled drug delivery on mucosal surfaces. | 2012 | J Control Release | pmid:21963767 |
| Onoue S et al. | Inhalable dry-emulsion formulation of cyclosporine A with improved anti-inflammatory effects in experimental asthma/COPD-model rats. | 2012 | Eur J Pharm Biopharm | pmid:22008148 |
| Cohen-Avrahami M et al. | Penetratin-induced transdermal delivery from H(II) mesophases of sodium diclofenac. | 2012 | J Control Release | pmid:22306174 |
| Lopes I et al. | Toxicity and genotoxicity of organic and inorganic nanoparticles to the bacteria Vibrio fischeri and Salmonella typhimurium. | 2012 | Ecotoxicology | pmid:22314390 |
| Garti N et al. | Lipolysis and structure controlled drug release from reversed hexagonal mesophase. | 2012 | Colloids Surf B Biointerfaces | pmid:22341515 |
| Issa JP et al. | Biological evaluation of the bone healing process after application of two potentially osteogenic proteins: an animal experimental model. | 2012 | Gerodontology | pmid:22970792 |
| Labahn J et al. | Vapor diffusion-controlled meso crystallization of membrane proteins. | 2012 | Methods Mol. Biol. | pmid:22976020 |